 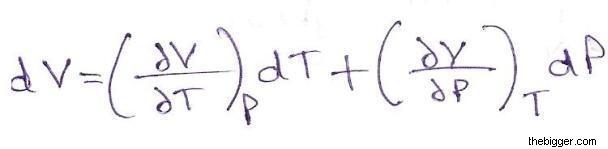
For an ideal gas, this means the volume of a gas is proportional to its temperature (historically, this is called Charles' law ). We will discuss isothermal process in a subsequent Atom.Īn isobaric process is a thermodynamic process in which pressure stays constant: ΔP = 0. This Atom addresses isobaric process and correlated terms. For example, an ideal gas that expands while its temperature is kept constant (called isothermal process) will exist in a different state than a gas that expands while pressure stays constant (called isobaric process). Under a certain constraint (e.g., pressure), gases can expand or contract depending on the type of constraint, the final state of the gas may change. specific heat: The ratio of the amount of heat needed to raise the temperature of a unit mass of substance by a unit degree to the amount of heat needed to raise that of the same mass of water by the same amount.the first law of thermodynamics: A version of the law of energy conservation: the change in the internal energy of a closed system is equal to the amount of heat supplied to the system, minus the amount of work done by the system on its surroundings.Conversely, if the environment does work on the system so that its internal energy increases, the work is counted as negative (for details on internal energy, check our Atom on "Internal Energy of an Ideal Gas"). As this work is done by using internal energy of the system, the result is that the internal energy decreases. If, for example, the system expands by a piston moving in the direction of force applied by the internal pressure of a gas, then the work is counted as positive. 
īy convention, work is defined as the work the system does on its environment. PV = 1 3 Nm v 2 ‾ \text W _ A → B = ∫ _ V _ A V _ B p dV = ∫ _ V _ A V _ B V NkT dV = NkT ln V _ A V _ B . 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
